You’re counting that study twice!
Why study grouping breaks meta-analyses (and how otto-SR fixes it)
By Christian Cao | February 16, 2026
TL;DR
- Systematic reviews analyze “studies.” But studies can be reported through many different papers/reports. Misidentifying which papers report on the same study can distort the final results.
- Otto can automatically group papers reporting on the same underlying study, saving researchers several hours of challenging work.
- Our algorithm caught several potential errors that impact downstream meta-analysis
When performing a systematic review, it’s common to encounter different papers all talking about the same underlying dataset (‘study’).
For example, a single cohort from a trial may be described in a conference abstract, a preprint, a published paper, and a later follow-up analysis.
- If researchers don’t properly group these different papers reporting on the same study, each paper might get analyzed, and the same trial can be counted twice (or more) in the final analysis.
- Alternatively, if researchers incorrectly group different papers as a single study, they can unintentionally exclude eligible studies from downstream analysis.
Both errors distort sample sizes, bias effect estimates, and can meaningfully change conclusions.
Consequently, researchers must carefully identify and group related reports into “study groups” before performing data analysis. This is a tedious and difficult task, and can take hours to days depending on the size and complexity of the review.
With Otto, we can automatically identify and group these related reports with near-perfect recall and precision.
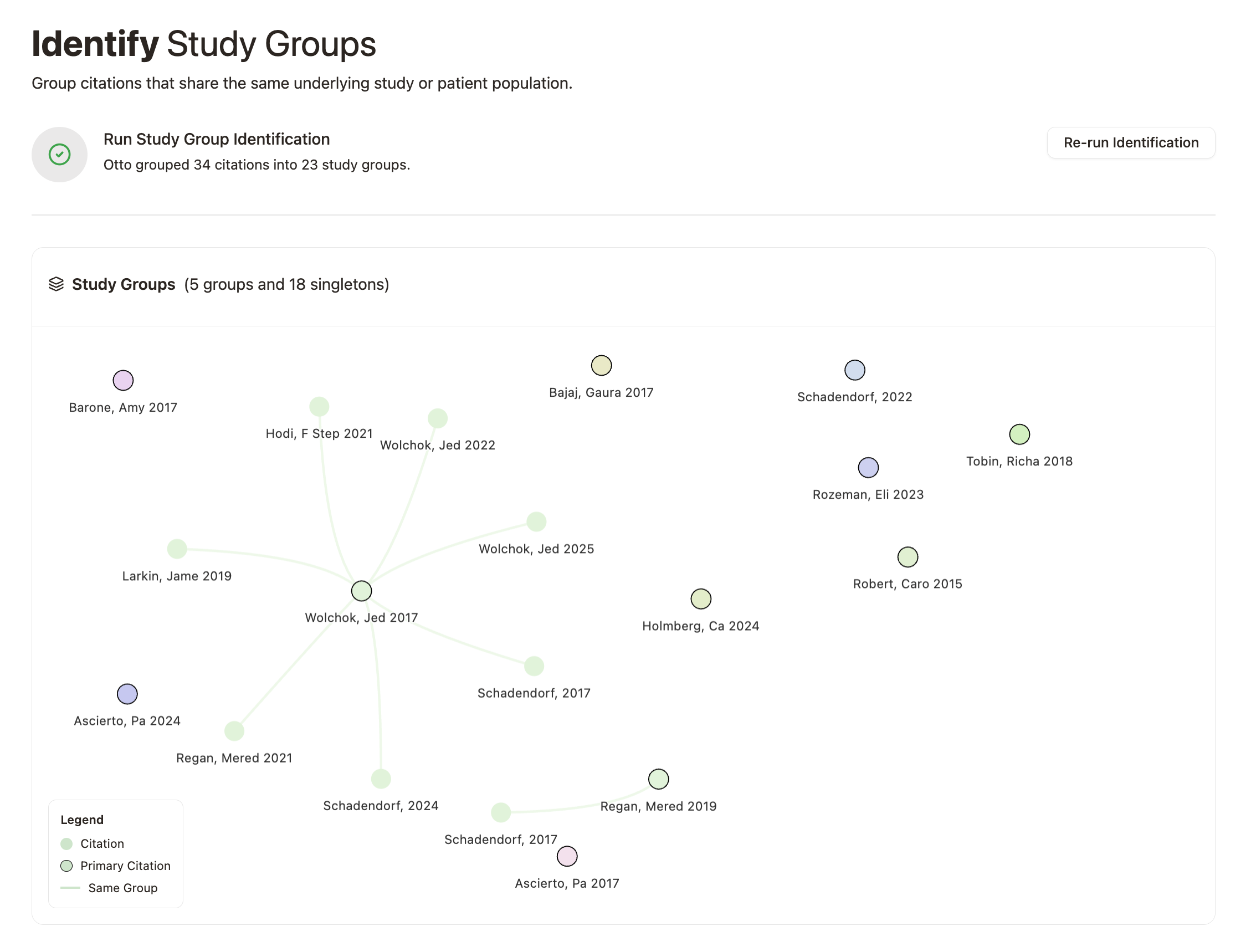
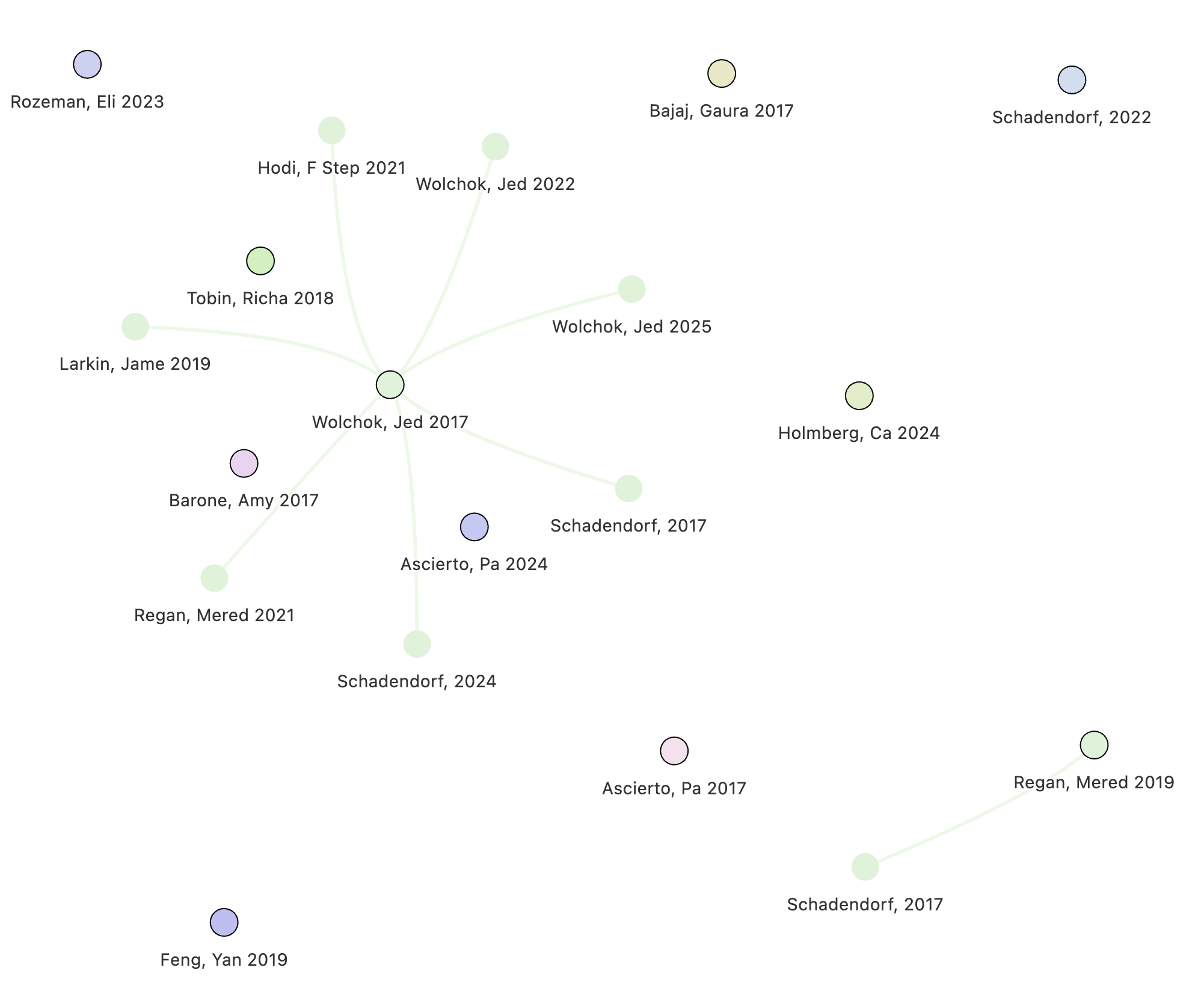
Study groups in Cochrane Reviews
To benchmark the performance of our algorithm, we analyzed a random sample of 100 Cochrane reviews.
Cochrane reviews represent the best evidence practices available.[1] They have rigorous reporting requirements and authors carefully document how multiple reports are linked to underlying study groups.
However, even in these high-quality reviews, our algorithm identified instances where study-grouping decisions could reasonably be reconsidered.
In our sample, we found that up to 3% of reviews contained study-grouping discrepancies that could impact final meta-analytic estimates, meaning that either
- Researchers grouped reports that were not related (false exclusion); or
- Researchers failed to group papers that were related (over-counting)
Let’s explore some examples below!
Hormone therapy for sexual function in perimenopausal and postmenopausal women (n = 218 citations)
We identified a few potential errors in the Cochrane review: “Hormone therapy for sexual function in perimenopausal and postmenopausal women” (Lara 2023).
Diem 2018 and Mitchell 2018 are included in the following analysis:
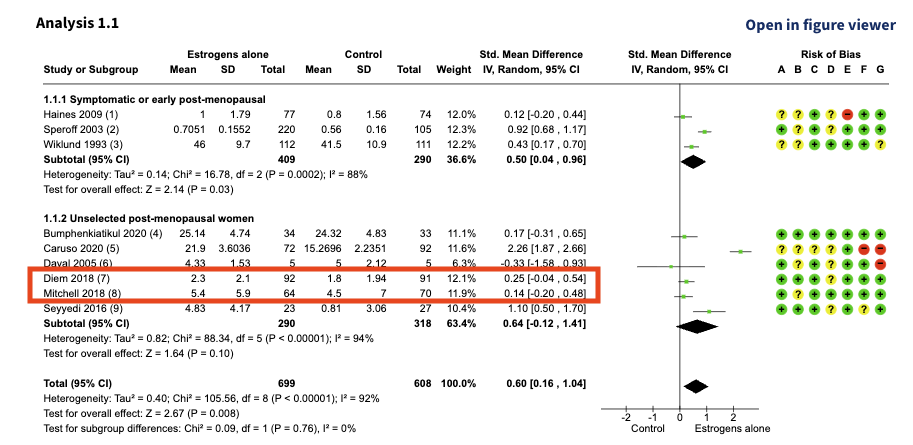
At first glance, Diem 2018 and Mitchell 2018 look like two separate studies. The sample sizes, mean values, and effect estimates are all different.
However, both papers cite the exact same clinical trial: NCT02516202, meaning that they aren’t two independent trials; they’re two reports of the same trial. The difference in values is because each paper reports a different questionnaire (or outcome) measuring a similar concept.
Diem 2018 reports outcomes using the MENQOL scale, while Mitchell 2018 reports outcomes using the FSFI index. Treating them as separate studies double counts their impact.
Nathorst-Böös 1993 and Wiklund 1993 were also included in the following analysis:
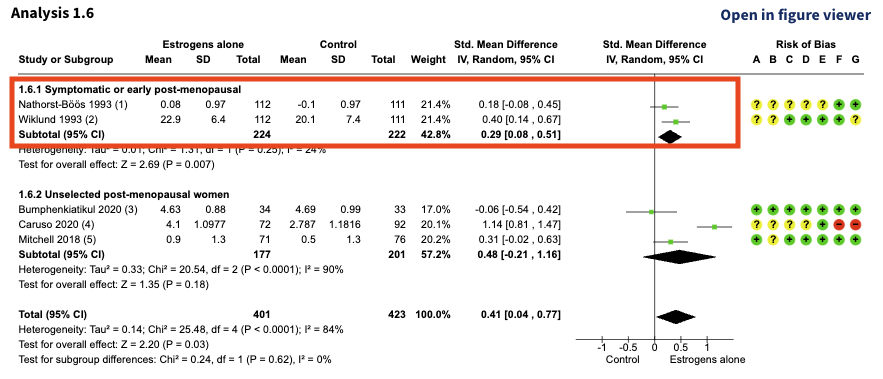
At first glance, Nathorst-Böös 1993 and Wiklund 1993 also look like two separate studies. The sample sizes are the same, but the mean values and effect estimates are different.
Let’s take a closer look:
| Study Name | Population | Interventions | Outcomes | Sample Details |
|---|---|---|---|---|
| Nathorst-Böös 1993 | Postmenopausal women between 45 and 65 years of age requiring hormonal replacement therapy (HRT). Recruited from 15 centers located in different parts of Sweden. | Estradiol therapy (Estraderm 50 ug/24 h) (E) or Placebo (P) | “A Swedish version of ‘McCoy’s Sex Scale Questionnaire’” with nine items | 242 women were randomised; n = 112 E; 111 P. 3 participants “who did not fulfil the inclusion criteria were excluded from the analysis” |
| Wiklund 1993 | Postmenopausal women between 45 and 65 years old requiring hormone replacement therapy for climacteric symptoms. Women from 15 centers volunteered for the study. | Transdermal estradiol therapy, 50 ug/24 hours, or placebo given as patches twice a week | McCoy Sex scale. “An abbreviated form with nine items was used” | 242 women were randomized; n = 112 E; 111 P. “Three of these were excluded from the analysis as protocol violators” (not fulfilling the inclusion criteria?) |
These two studies seem suspiciously similar. They have identical populations, interventions, outcomes, and study characteristics.
So why are the reported means different?
- Nathorst-Böös 1993 analyzed a single question from the McCoy scale (Question 8).
- Wiklund 1993 analyzed the aggregate of three questions from the scale.
In other words, the outcome definitions differ, but the same instrument was used. However, both reports were incorrectly treated as independent studies in the meta-analysis. Again, the same trial was counted twice.
Interventions for preventing posterior capsule opacification (n = 322 citations)
In the Cochrane review: “Interventions for preventing posterior capsule opacification” (Findl 2010), Hayashi 1998 was used in the following analysis:
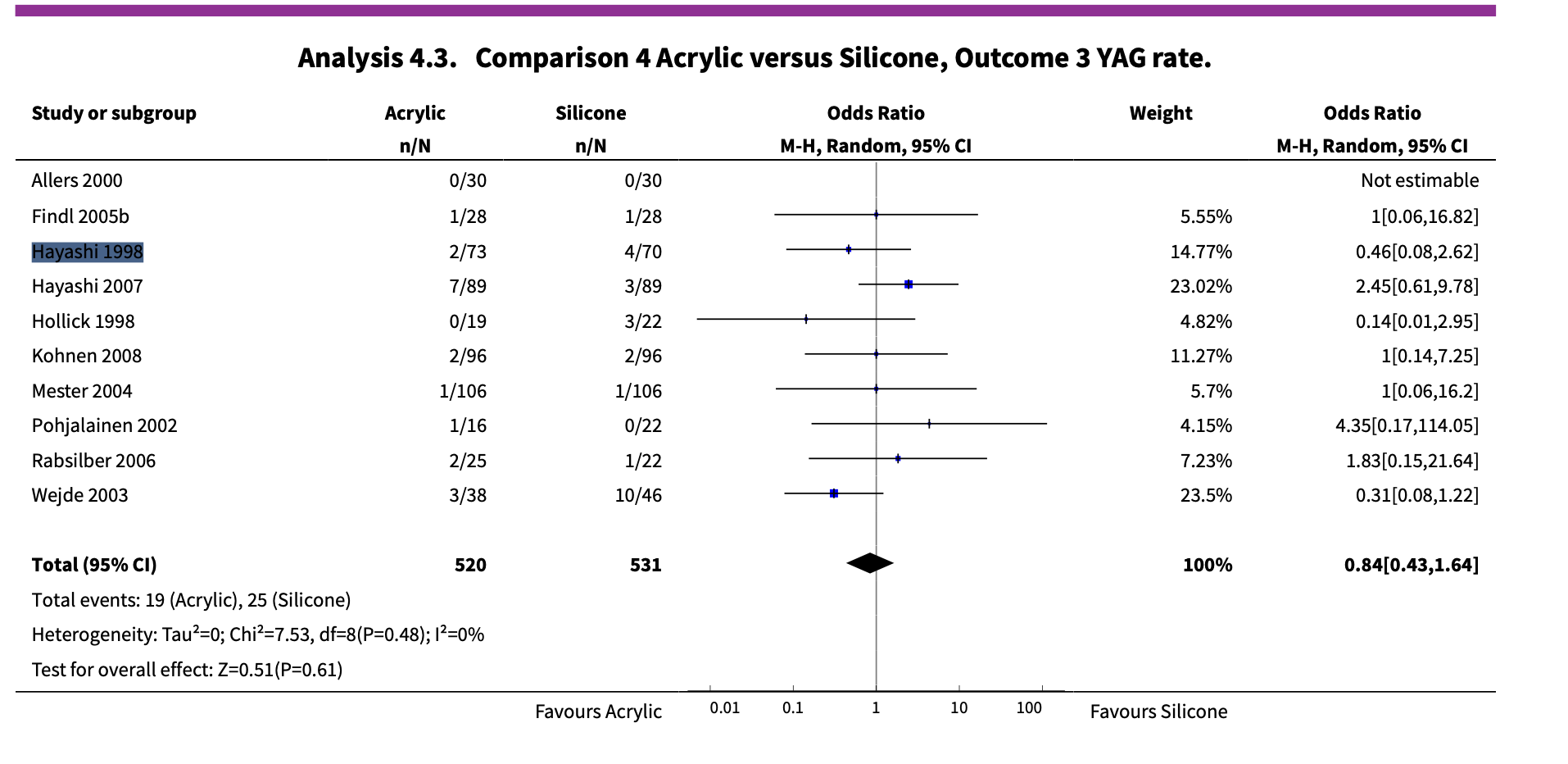
The authors group the Hayashi 1998 study from two papers:
- Hayashi’s original 1998 paper “Quantitative comparison of posterior capsule opacification after polymethylmethacrylate, silicone, and soft acrylic intraocular lens implantation”; and,
- Hayashi’s 2001 paper “Changes in posterior capsule opacification after poly(methyl methacrylate), silicone, and acrylic intraocular lens implantation”.

At first glance, the studies seem to be the same, sharing similar author lists and titles—maybe it’s a follow-up study?
The reporting in Hayashi 1998 also lacks detail; the methods don’t provide a clear explanation for where the patients came from.
However, upon closer inspection, these might not be two reports of the same patient cohort, they could be distinct studies.
Our system caught a small nuance buried in the discussion section of Hayashi 2001. Here, Hayashi 2001 cited Hayashi 1998 as a “previous study” that was performed retrospectively in their discussion. Meanwhile, Hayashi 2001 is a prospective cohort. This gives us reason to suspect that Hayashi 1998 and Hayashi 2001 are actually two distinct studies that analyzed separate patient cohorts.
Unfortunately, the authors only extract outcome data from the Hayashi 1998 paper; data from Hayashi 2001 was not considered in any meta-analysis. This means that the Cochrane review had incorrectly excluded an eligible study in their meta-analysis.
Aspirin for in vitro fertilisation (n = 157 citations)
In the Cochrane review: “Aspirin for in vitro fertilisation” (Siristatidis 2016), Haapsamo 2010 and Pakilla 2005 were analyzed as follows:
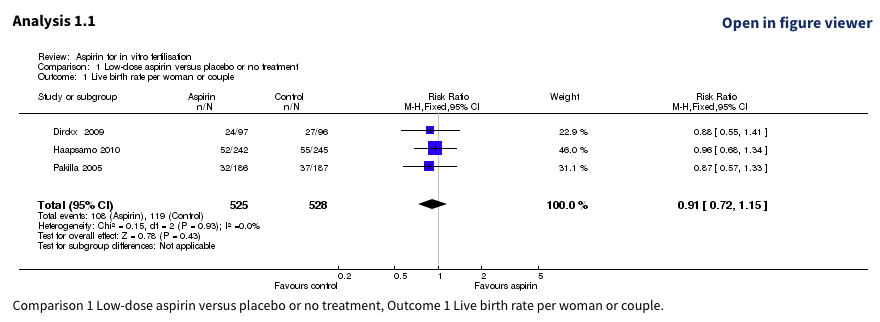
Let’s take a closer look at each study within the study group:
| Study Name | Population | Intervention | Setting | Sample size |
|---|---|---|---|---|
| Pakilla 2005 | unselected IVF/ICSI patients, age <40, <4 prior stimulations | 100 mg aspirin daily vs placebo | Oulu University Hospital | 487 |
| Haapsamo 2010 | unselected IVF/ICSI patients, age <40, <4 prior stimulations | 100 mg aspirin daily vs placebo | Oulu University Hospital | 374 |
| NCT00683202 | unselected IVF/ICSI patients, age <40, <4 prior stimulations | 100 mg aspirin daily vs placebo | Oulu University Hospital | 374 |
Another tricky case! The study design appears similar, but the sample sizes don’t seem to line up.
Interestingly, Haapsamo 2010 directly acknowledges: “The study protocol has been previously described (Päkkilä et al., 2005)”, and cites the NCT00683202 trial.
Given the strong concordance between NCT00683202 and Pakilla 2005, it seems that Haapsamo 2010 may be an update to the trial described in Pakilla 2005 with further prospective enrolment.
Here, both reports were treated as independent studies in the meta-analysis. If Haapsamo 2010 is an update, then a large set of trial participants were counted twice!
otto-SR’s mission
The errors above weren’t made by careless reviewers; they were made by some of the best in the field, working within the most rigorous framework we have.
Our algorithm catches subtle distinctions: a “previous study” buried in a discussion section, a shared trial registration hidden behind different author lists, or the same patients concealed by different outcome measures.
If you’d like to see how Otto handles related reports in your reviews, get in touch. And if you’re interested in shaping the future of evidence synthesis, reach out to join us here!